Faculty Information

Amit B. Pawar
Office:
Personal Information
Professional Experience
Assistant Professor, IIT Mandi, (2019-)
DST-Inspire Faculty, CSIR-Indian Institute of Chemical Technology, Hyderabad, INDIA, 2015-2019
Postdoctoral Associate, Prof. Sukbok Chang's Group, KAIST, Daejeon, Republic of Korea, 2013-2015
Educational Background
Ph.D., Indian Institute of Science, Bangalore, 2007-2012 (With Prof. K. R. Prasad)
M.Sc. (Organic Chemistry) from University of Pune, Pune, 2005-2007
B.Sc. (Chemistry) from University of Pune, Pune, 2002-2005
Broad Area
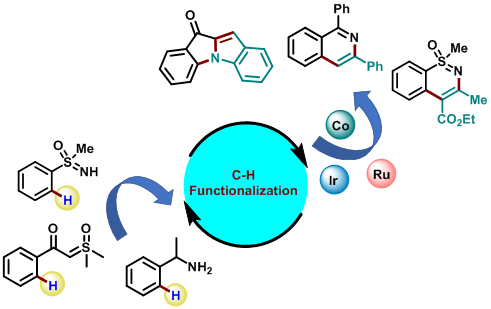
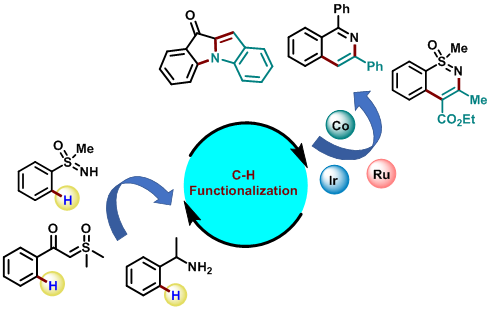
Organic Synthesis, C–H Activation
Specific Areas
Cp*Co(III)-Catalysed C-H Functionalization
Total Synthesis of Natural Products
Photo-redox Catalysis and Electrocatalysis
CY502-Photochemistry and Pericyclic Reactions
CY501-Organic Reactions and Mechanism
CY504-Heterocyclic Chemistry
DST-Inspire Faculty Award by DST, New Delhi, India (January 2015)
Received 1st prize from Eli-Lilly (USA) for "Eli Lilly Asia Outstanding Thesis Award 2012."
Senior Research Fellowship (SRF) by CSIR, New Delhi, India (2010- 2012).
Junior Research Fellowship (SRF) by CSIR, New Delhi, India (2007-2010).

Amit B. Pawar
Organic Synthesis
C–H Activation
First-row transition metals are available abundantly in Earth’s crust, compared to their 4d and 5d analogs. Despite their high natural abundance and low toxicity, first-row transition metals are underutilized in C-H activation. Our group is involved in the development of novel organic transformations using Cp*Co(III)-catalysis. In recent years, the area of mild C−H activation has gained significant importance, as it allows to carry out the reactions at or below room temperature and in the absence of any oxidant or reductant. This results in the enhancement of the functional group tolerance of the reactions and ultimately leads to the application in the late-stage functionalization of complex biomolecules. Our group is focusing on the development of mild C−H activation using Cp*Ir(III) catalysis especially using α-diazocarbonyl compounds for the synthesis of various biologically important heterocyclic scaffolds. Our group is also interested in the total synthesis of indolizidinone types of natural products and their analogous using C−H functionalization strategy.
- Free Amine-Directed Ru(II)-Catalyzed Redox-Neutral [4 + 2] C−H Activation/Annulation of Benzylamines with Sulfoxonium Ylides. Yogesh N. Aher and Amit B. Pawar* J. Org. Chem 2022, doi.org/10.1021/acs.joc.2c00931.
- Cp*Co(III)-catalyzed C–H amination/annulation cascade of sulfoxonium ylides with anthranils for the synthesis of indoloindolones. Yogesh N. Aher and Amit B. Pawar* Chem. Commun. 2021, 57, 7164.
- Cp*Ir(III)-Catalyzed C−H/O−H Functionalization of Salicylaldehydes for the Synthesis of Chromones at Room Temperature. Dhanaji M. Lade, Yogesh N. Aher and Amit B. Pawar* J. Org. Chem. 2019, 84, 9188.
- Cp*Ir(III)-catalyzed C−H/N−H functionalization of sulfoximines for the synthesis of 1,2-benzothiazines at room temperature. Yogesh N. Aher, Dhanaji M. Lade and Amit B. Pawar* Chem. Commun. 2018, 54, 6288.
- Cp*Co(III)-catalyzed C−H functionalization cascade of N‑methoxyamides with alkynedione for the synthesis of indolizidines. Lahu N. Chavan; Krishna Kumar Gollapelli; Rambabu Chegondi* and Amit B. Pawar* Org. Lett. 2017, 19, 2186.
- 2023
-
-
Reverse Regioselective Cp*Co(III)-Catalyzed [4+2] C−H Annulation of N-Chloroamides with Vinylsilanes: Synthesis of 4-Silylated Isoquinolones and Their Synthetic Utilities, Arijit Ghosh^, Tamanna Rana^, Nilanjan Bhaduri, and Amit B. Pawar*, Org. Lett. 2023, 25, 7878. (^Contributed equally)
- Harnessing Vinyl Acetate as an Acetylene Equivalent in Redox-Neutral Cp*Co(III)-Catalyzed C–H Activation/Annulation for the Synthesis of Isoquinolones and Pyridones. Tamanna Rana, Arijit Ghosh, Yogesh N. Aher, and Amit B. Pawar* ACS Omega 2023, 8, 28, 25262.
-
Advances in Transition Metal-Catalyzed C-H Amination Strategies using Anthranils. Yogesh N. Aher, Nilanjan Bhaduri, and Amit B. Pawar*, Org. Biomol. Chem. 2023 (DOI: 10.1039/d3ob01421e)
-
Redox-Neutral C−H Annulation Strategies for the Synthesis of Heterocycles via High-Valent Cp*Co(III)-Catalysis. Nilanjan Bhaduri and Amit B. Pawar*, Org. Biomol. Chem. 2023, 21, 3918.
-
Ru(II)-Catalyzed Regioselective Redox-Neutral [4 + 2] Annulation of N-Chlorobenzamides with 1,3-Diynes at Room Temperature for the Synthesis of Isoquinolones. Arijit Ghosh, Goraksha T. Sapkal, and Amit B. Pawar*, J. Org. Chem. 2023, 88, 4704.
-
- 2022
-
- Free Amine-Directed Ru(II)-Catalyzed Redox-Neutral [4 + 2] C−H Activation/Annulation of Benzylamines with Sulfoxonium Ylides. Yogesh N. Aher and Amit B. Pawar* J. Org Chem. 2022, 57, doi.org/10.1021/acs.joc.2c00931.
- Cp*Co(III)-Catalyzed C–H Functionalization Mediated by Oxidising Directing Groups Towards the Synthesis of Heterocycles. Yogesh N. Aher, Bhaskar Mondal* and Amit B. Pawar* Handbook of C-H Functionalization, Wiley-VCH: Weinheim, 2022.
- 2021
-
- Cp*Co(III)-catalyzed C–H amination/annulation cascade of sulfoxonium ylides with anthranils for the synthesis of indoloindolones. Yogesh N. Aher and Amit B. Pawar* Chem. Commun. 2021, 57, 7164.
- Synthesis of the macrolactone core of the revised structure of palmerolide C. Kannan Vaithegi, Amit B. Pawar, Kavirayani R. Prasad* Tetrahedron 2021,77, 131768.
- 2020
-
- 2019
-
- Catalyst- and reagent- free 1,6-hydrophosphonylation of p-quinone methides: A practical approach for the synthesis of diarylmethyl phosphine oxides. Yogesh N. Aher; Amit B. Pawar* Org. Biomol. Chem. 2019, 17,7536.
- Cp*Ir(III)-catalyzed C−H/O−H functionalization of salicylaldehydes for the synthesis of chromones at room temperature. Dhanaji M. Lade, Yogesh N. Aher, Amit B. Pawar* J. Org. Chem. 2019, 84, 9188.
- 2018
-
- Cp*Ir(III)-catalyzed C−H/N−H functionalization of sulfoximines for the synthesis of 1,2-benzothiazines at room temperature. Aher, Y. N.; Lade, D. M.; Amit B. Pawar* Chem. Commun. 2018, 54, 6288.